Ranexa is indicated as add-on therapy for stable angina in adults inadequately controlled by or intolerant to first-line therapies (beta-blockers and/or calcium antagonists).1

Neutral hemodynamic profile:No significant changes in blood pressure or heart rate.1
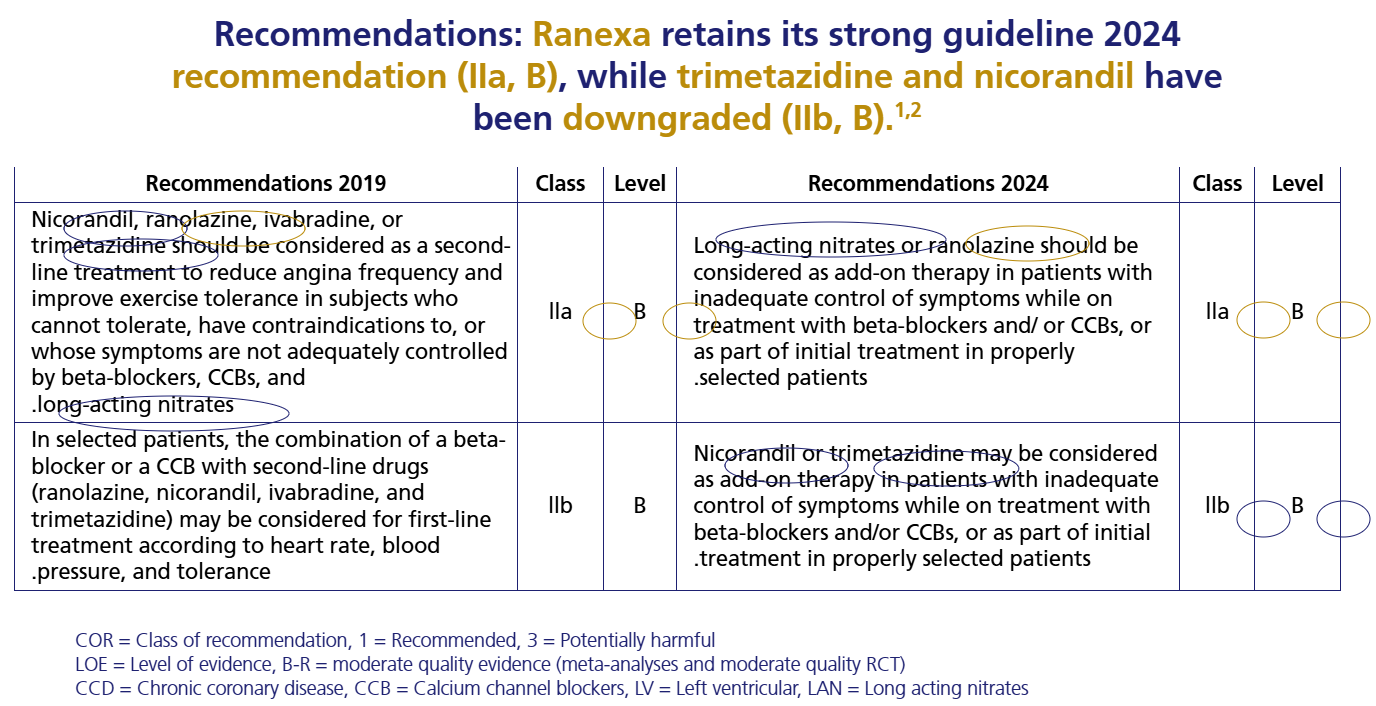
Guideline Recommendation

Reduced anginal episodes,
lower nitroglycerin use.6

Strong recommendation retained.7

Neutral hemodynamic profile:No significant changes in blood pressure or heart rate.1
Guideline Recommendation

Reduced anginal episodes,
lower nitroglycerin use.6

Strong recommendation retained.7

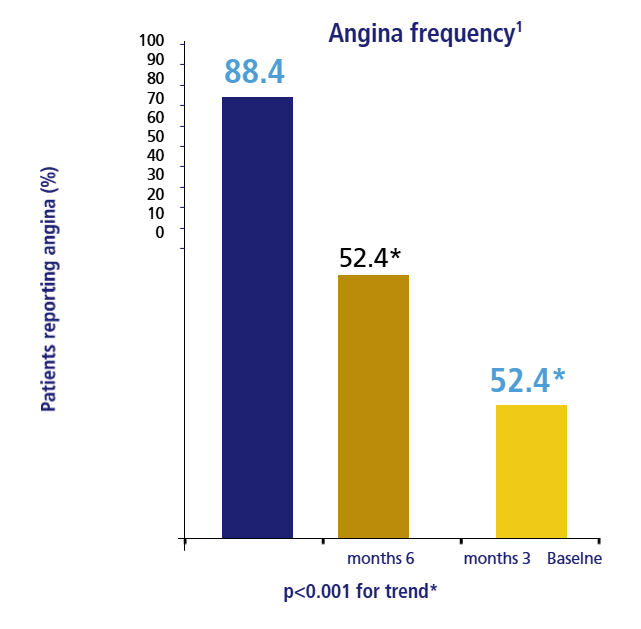
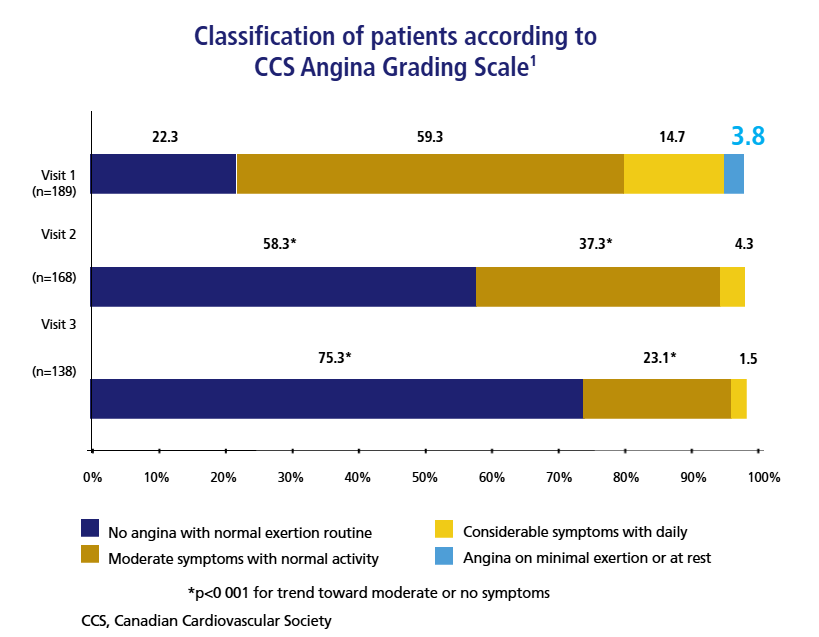
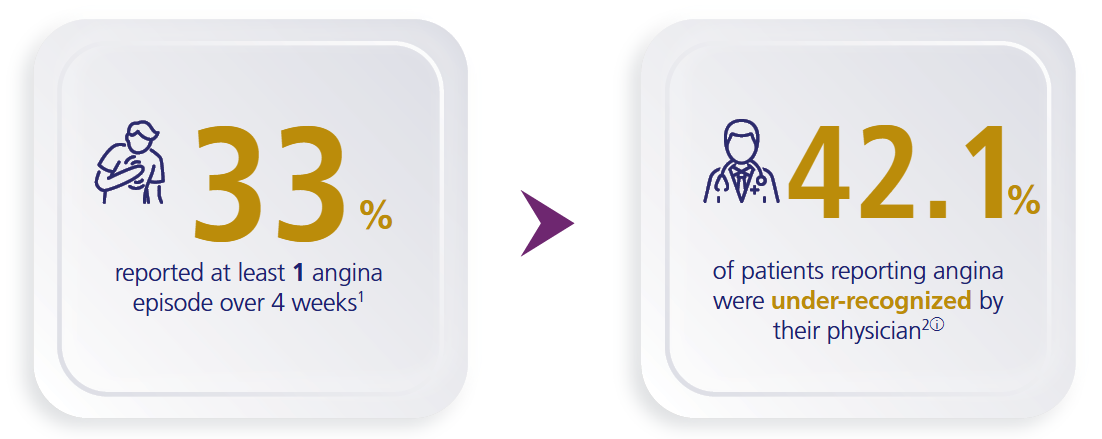
Under-recognition of angina by physicians could affect patients’ quality of life1
APPEAR Study: Among 1257 patients with coronary artery disease

Learn more about:
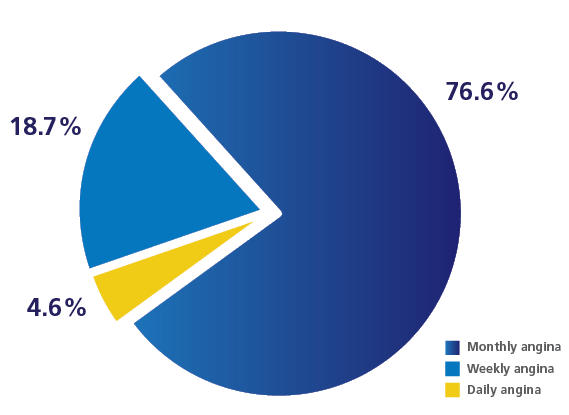
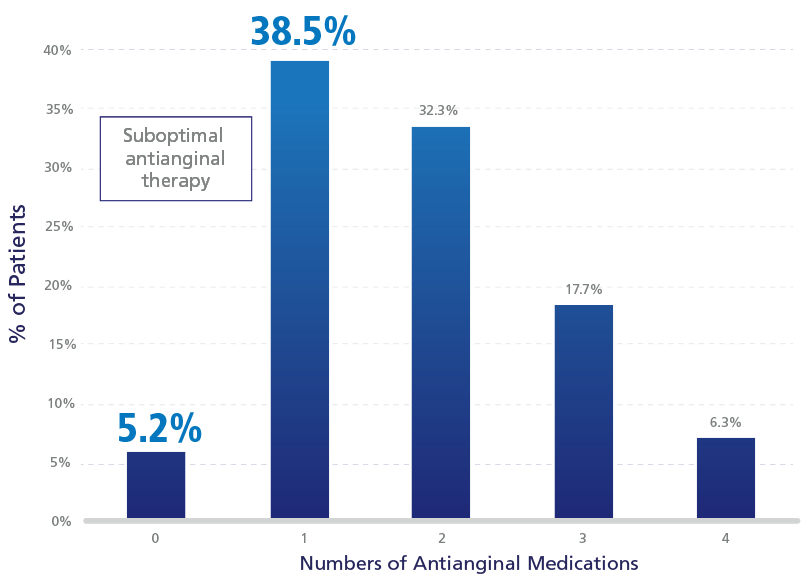
Frequency Distribution of Patient-Reported Angina

APPEAR study: 44% of patients with daily/weekly angina were on suboptimal antianginal therapy.1

For more details

:Key CCS Management Guidelines 2024 Takeaways


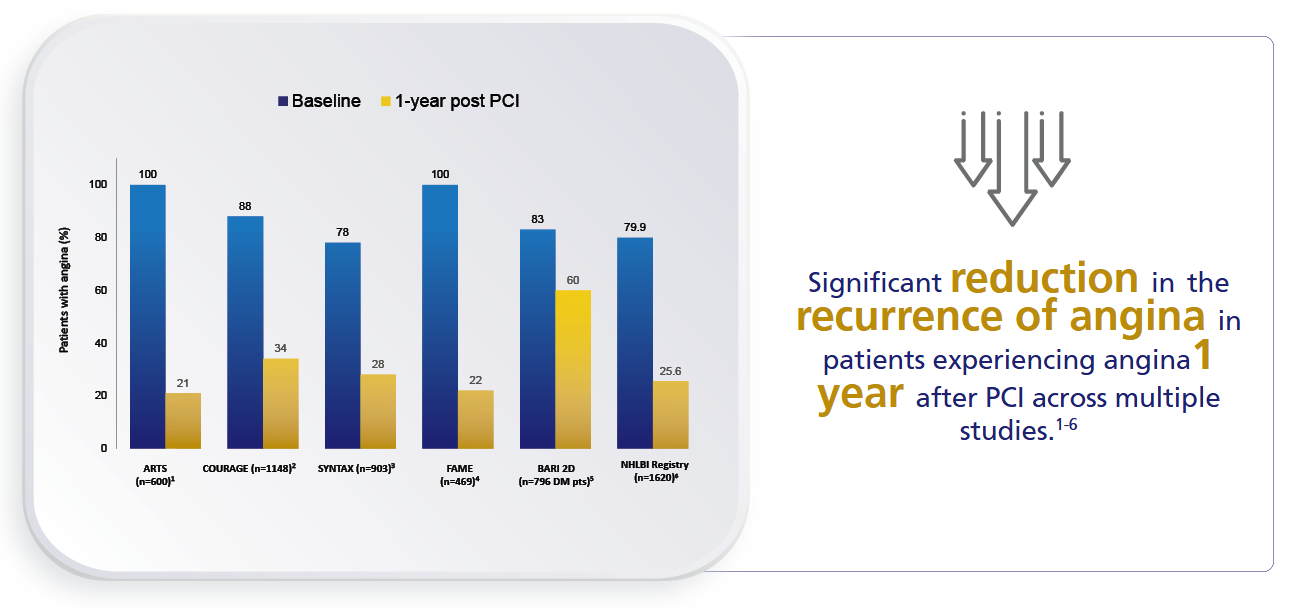
Recurrence of Angina After PCI1-6

Learn more about: The potential reasons for
persistent angina

Ranexa Patient Profiles



Ranexa: Redefine Life for Your Stable Angina Patients

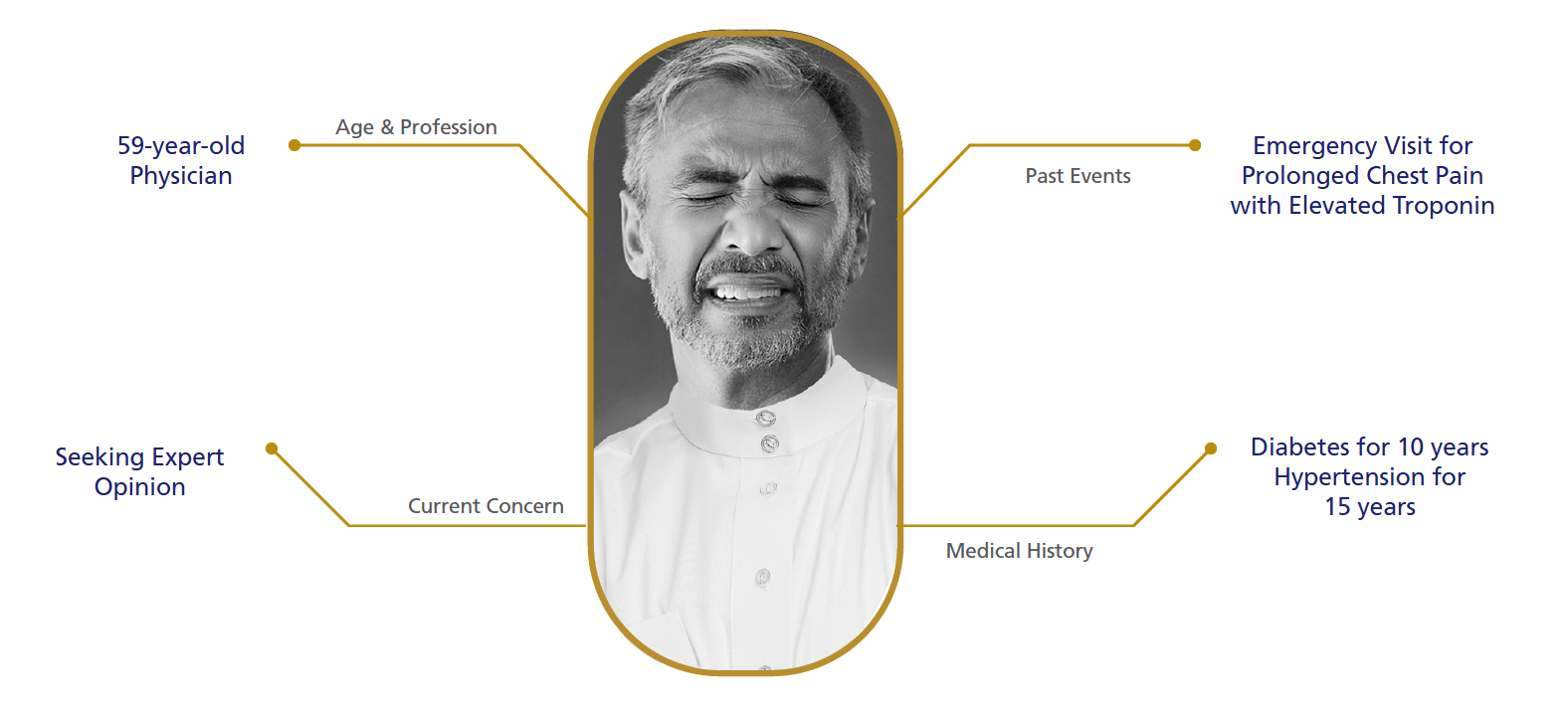
Diabetic Patient with CSA - History

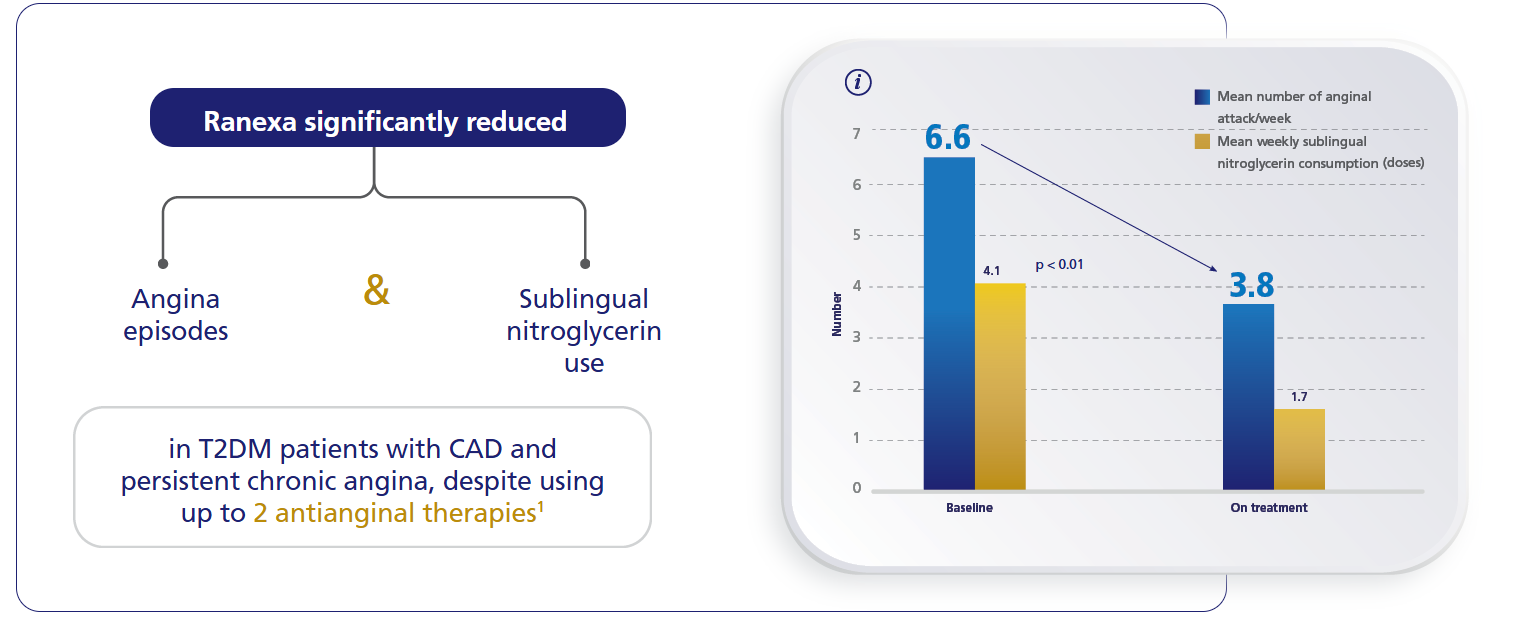
TERISA study1

For more details

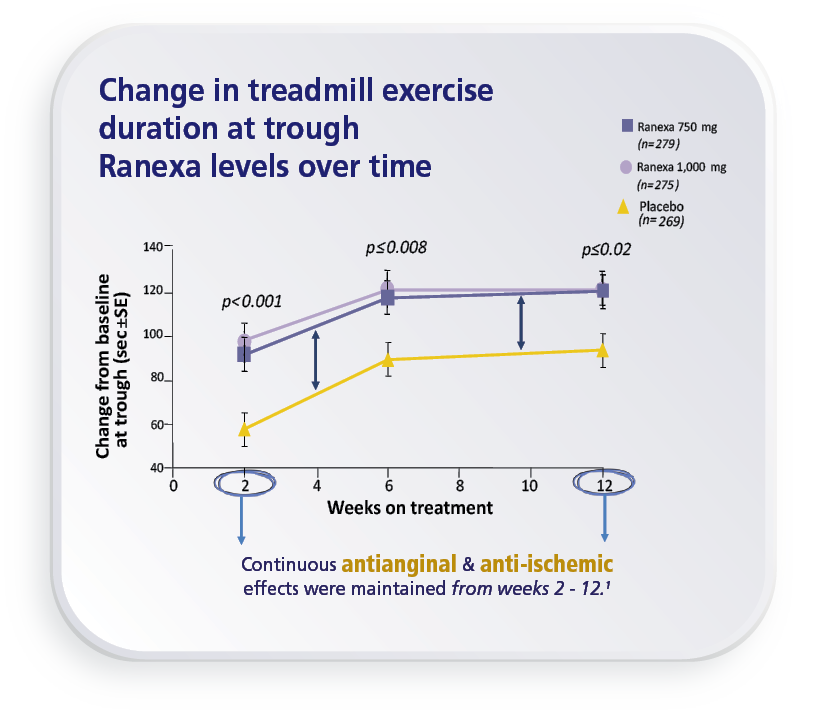
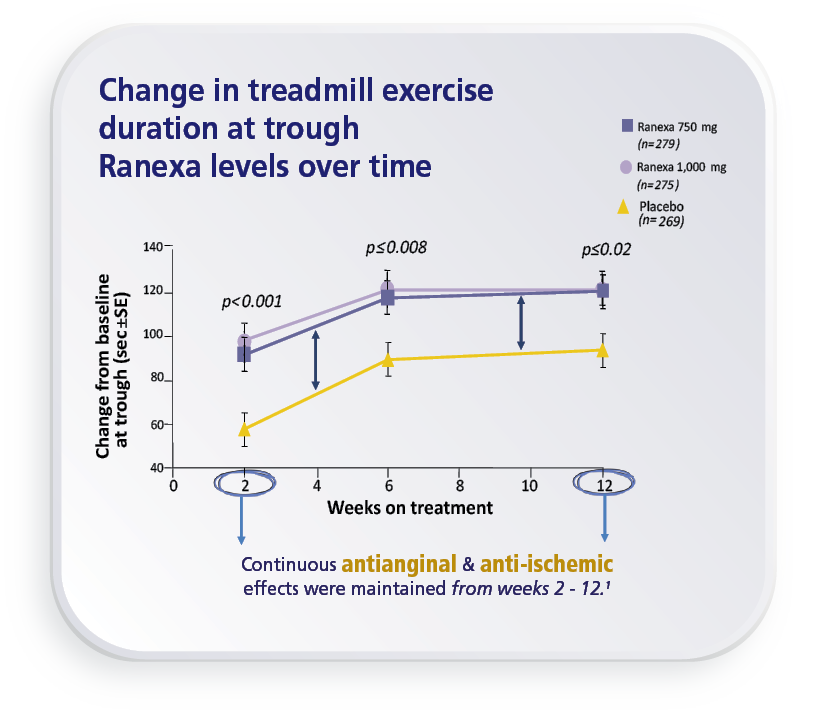
CARISA Study1

Ranexa significantly increased
treadmill exercise duration by 46.3 Sec at peak drug levels.1 (p=0.006)

For more details

CARISA Study1

Ranexa significantly increased
treadmill exercise duration by 46.3 Sec at peak drug levels.1 (p=0.006)


For more details

Diabetic Patient with CSA - History

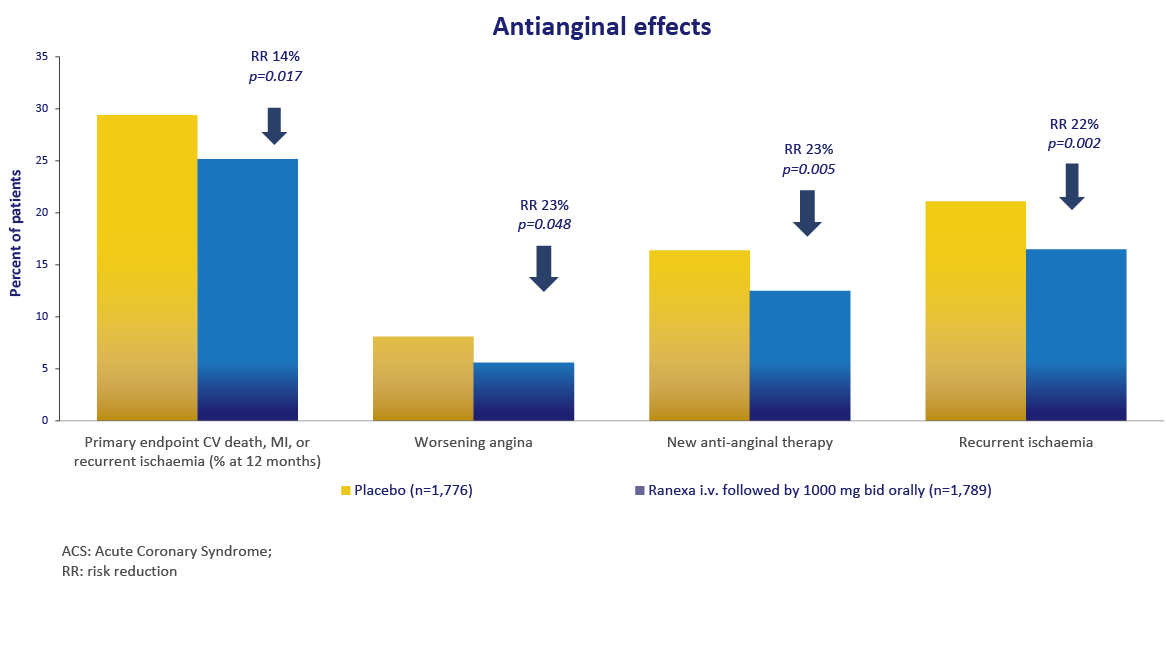
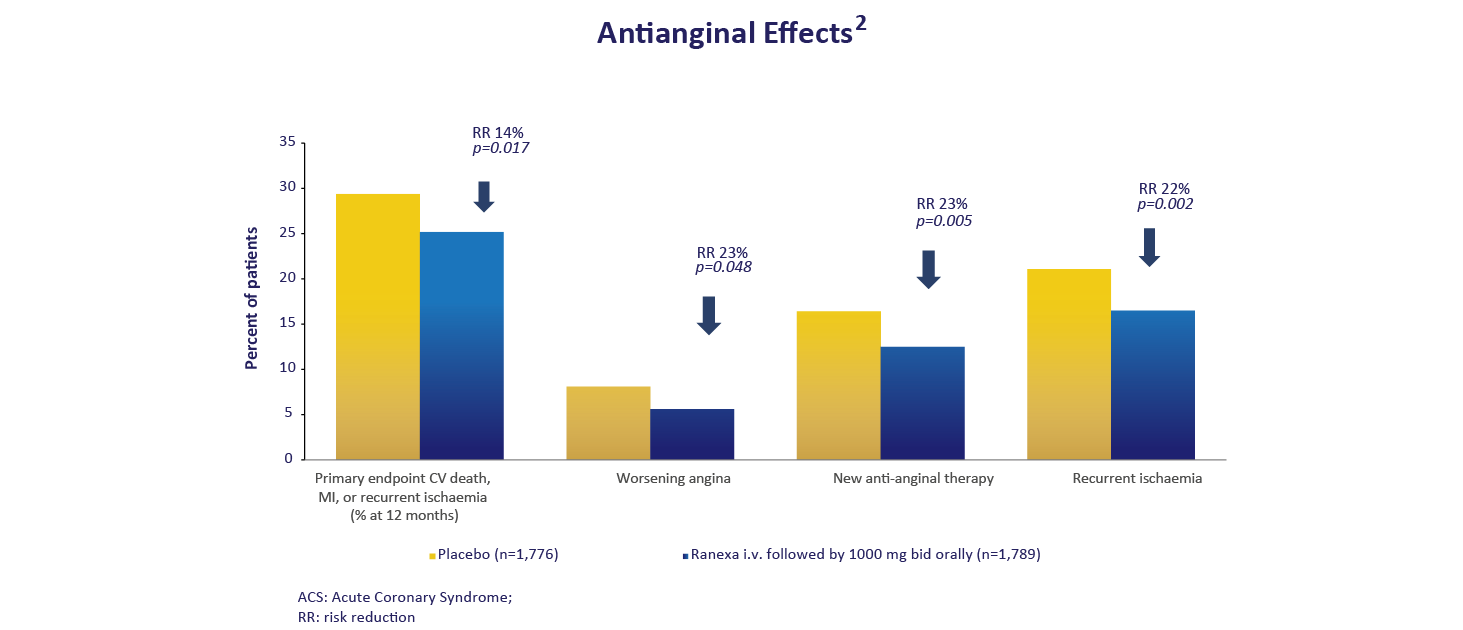
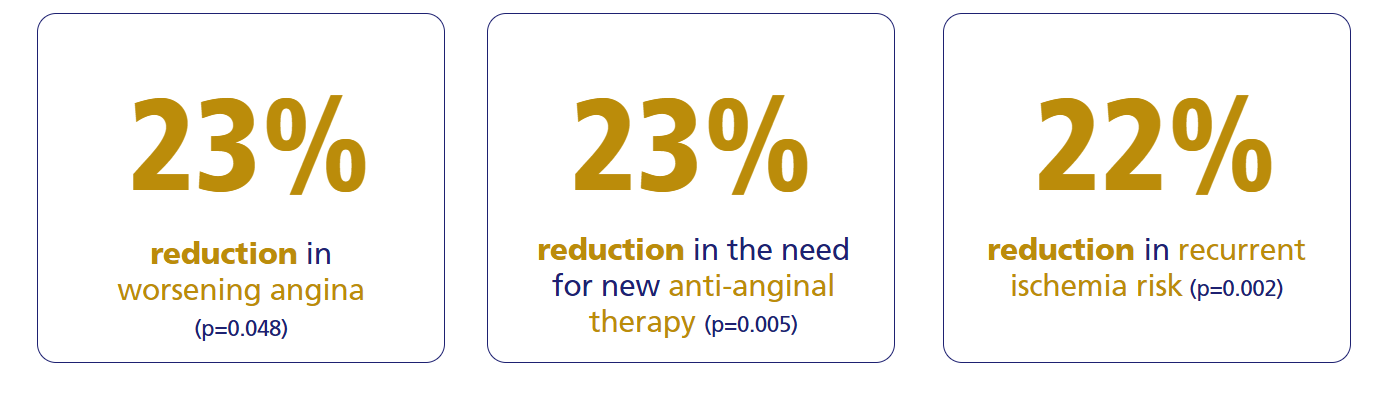
MERLIN-TIMI 36 Study: Ranexa’s Efficacy in Acute
Coronary Syndrome & Chronic Angina1,2

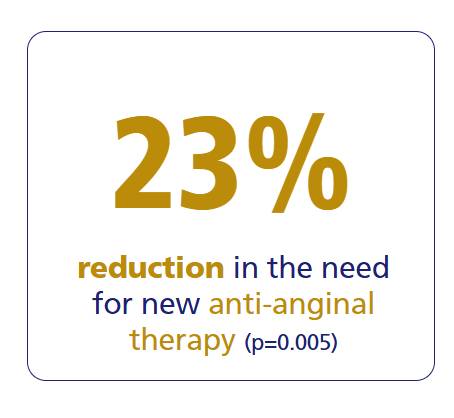

Ranexa significantly reduces cardiovascular endpoints and angina-related events compared to placebo.

For more details

MERLIN-TIMI 36 Study1

Ranexa significantly improved all measures of exercise performance among the patients with ACS and prior chronic angina.1
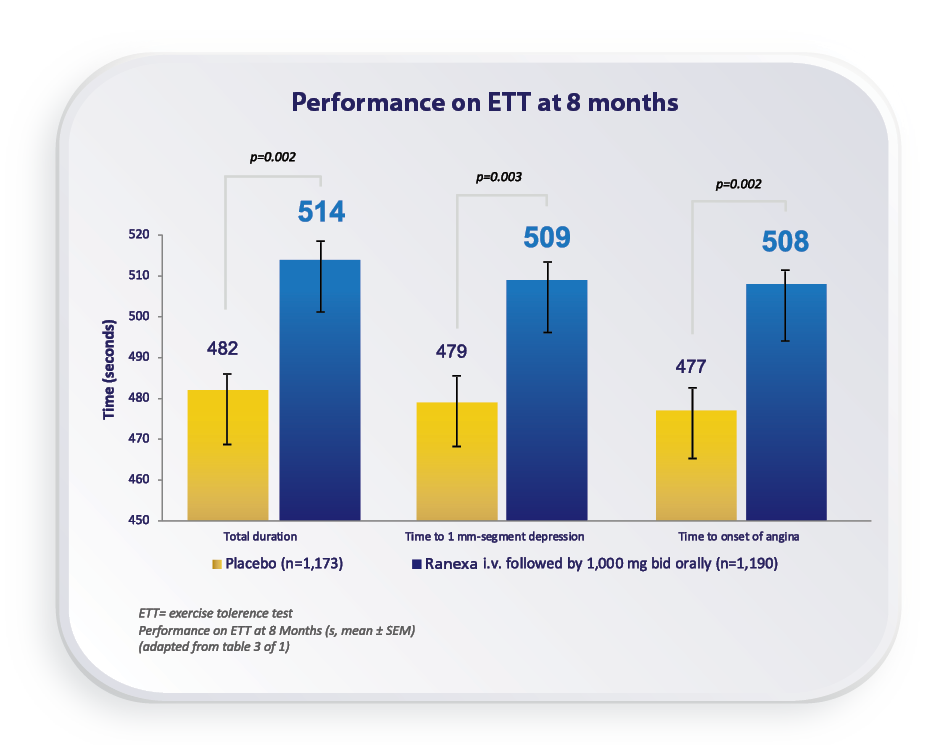

For more details

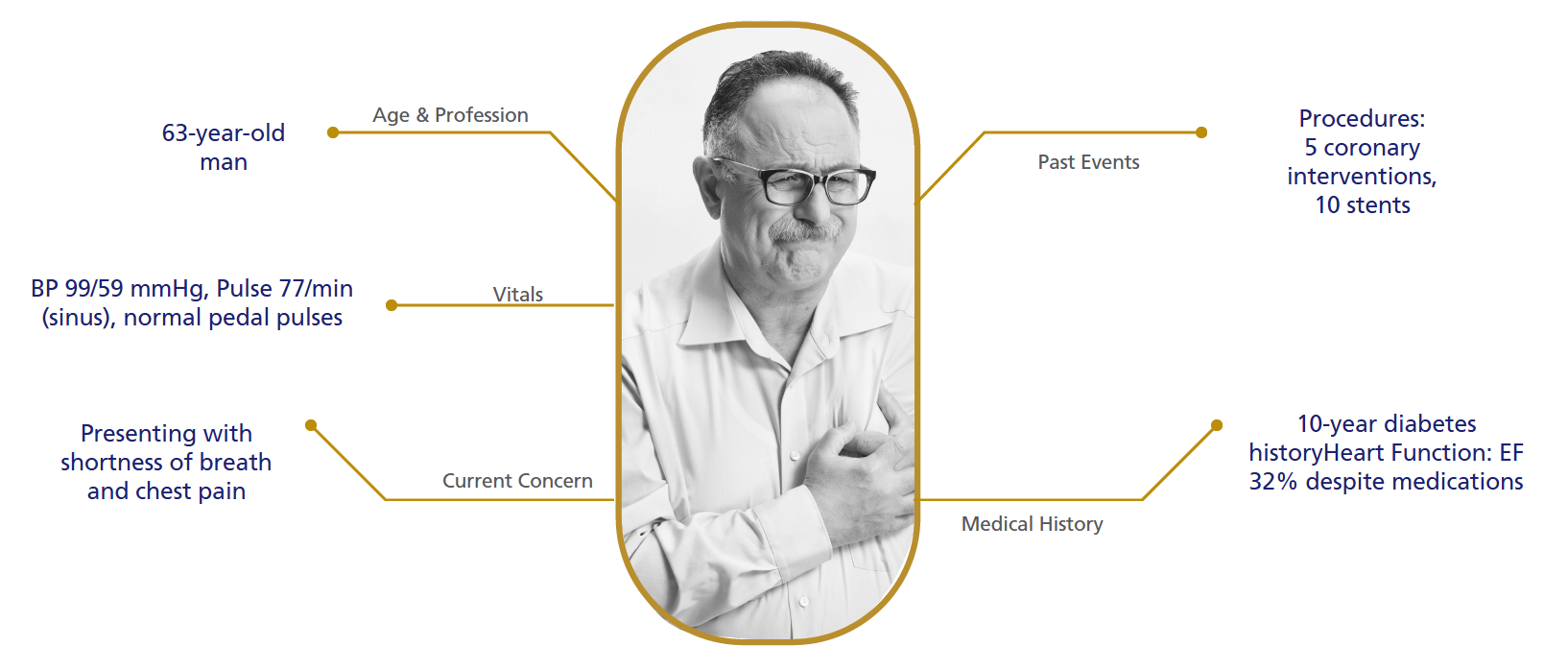
Symptomatic Patient on Beta Blockers - History
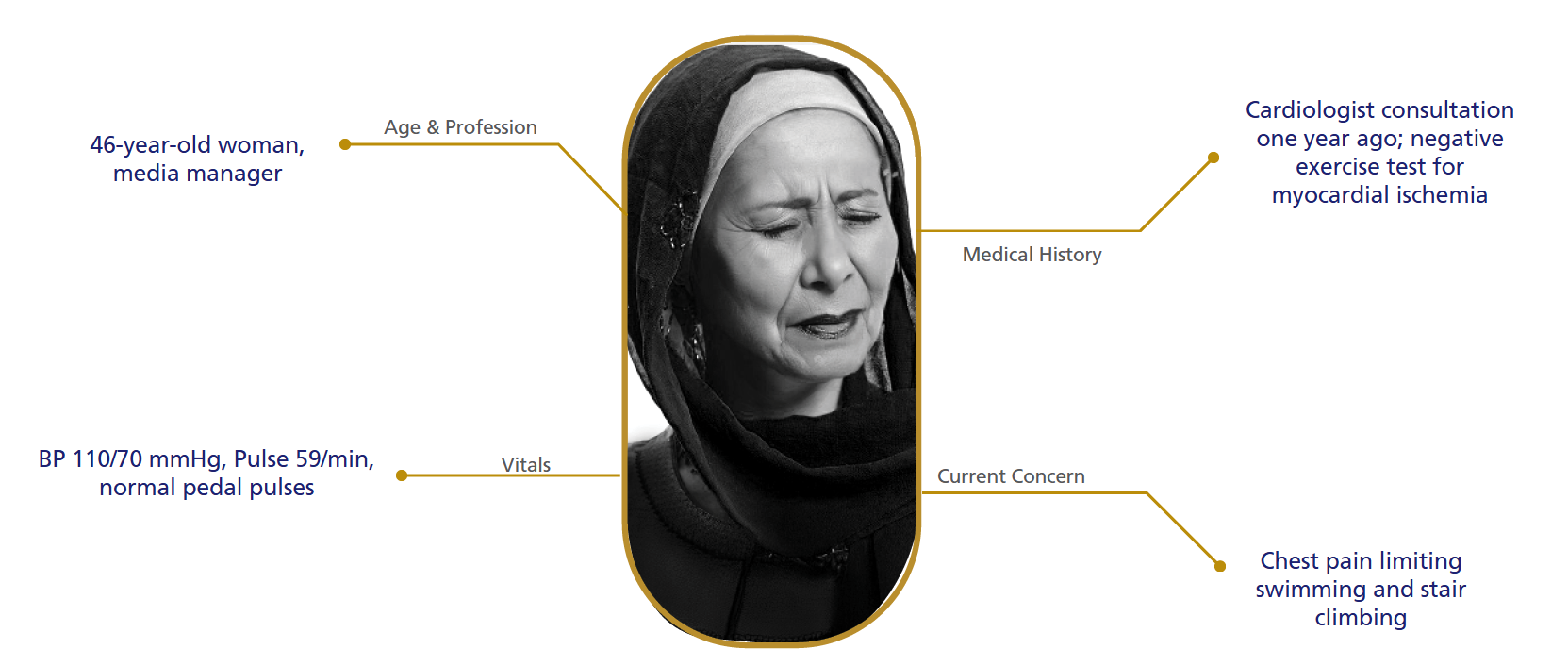

Ranexa Enhances Quality of Life and Symptom Control in Patients with Microvascular Angina.1
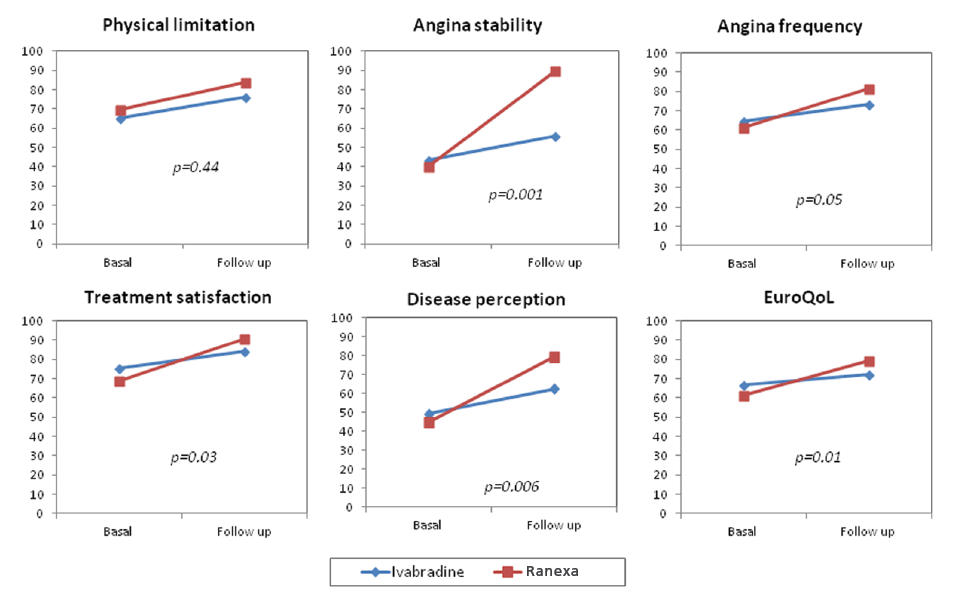

For more details

Ranexa Enhances Physical Functioning and Quality of Life1
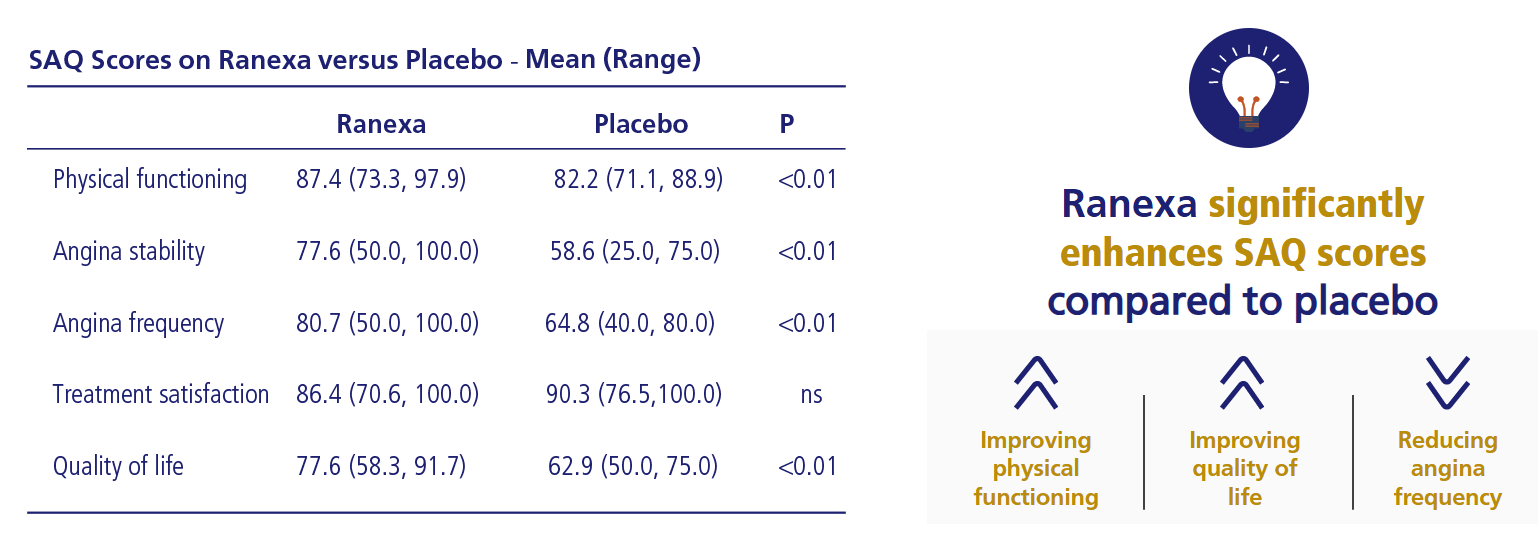

Study details

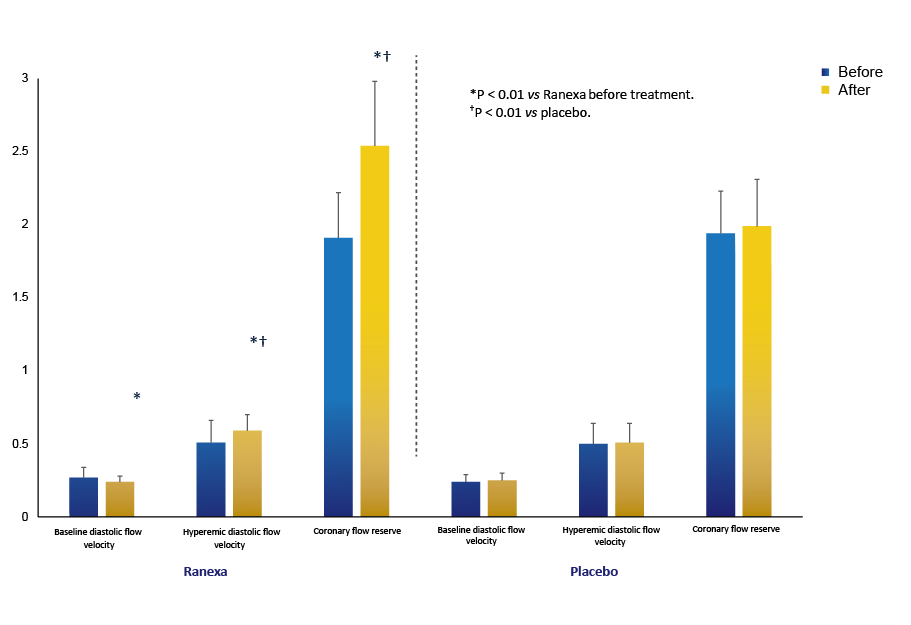
Ranexa Improves Coronary Flow Reserve
in Microvascular Angina1
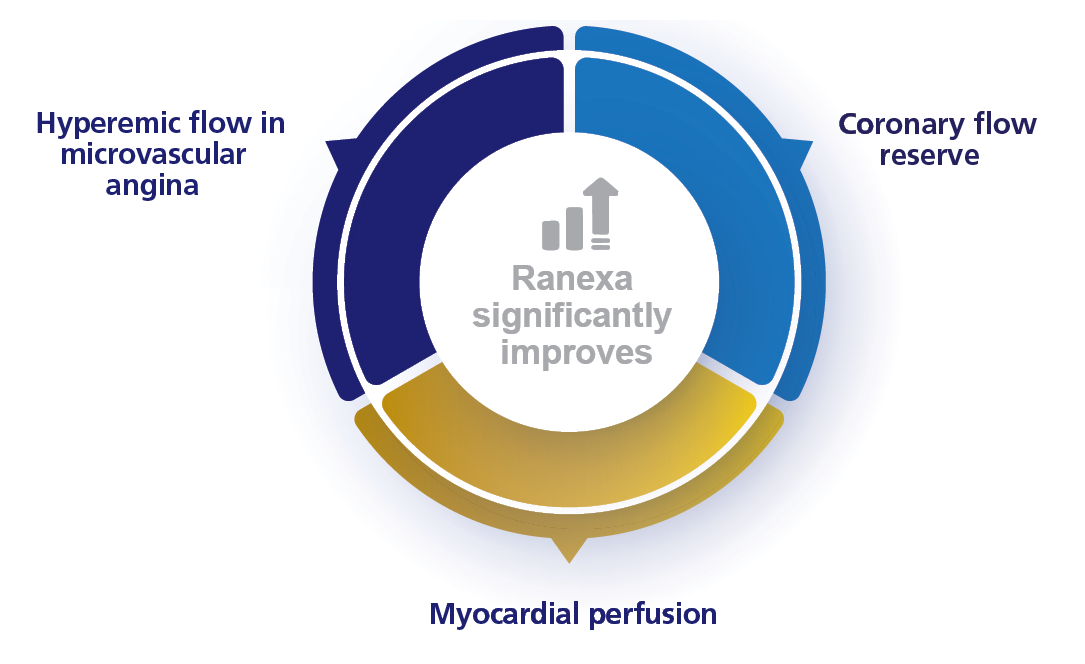

For more details

Ranexa’s Enhances Coronary Microvascular Function in Nonobstructive CAD1
Ranexa improves
3 of the 5 SAQ subscales:


Study details

Ranexa’s Enhances Coronary Microvascular Function in Nonobstructive CAD1
Ranexa improves
3 of the 5 SAQ subscales:


Study details

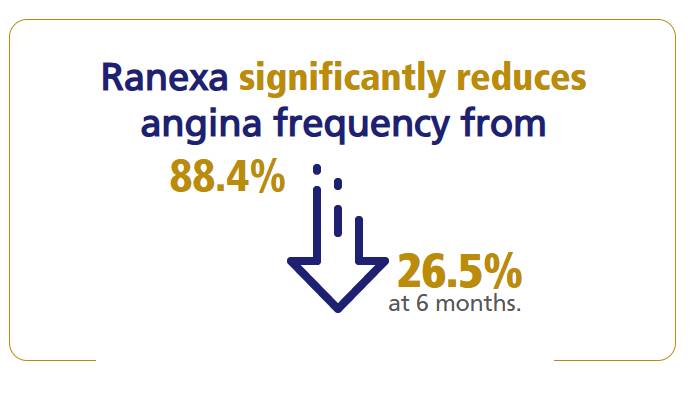
Efficacy of Ranexa



ERICA Study1

Ranexa provided significant additional antianginal benefit in patients who remained symptomatic despite maximum recommended therapy with a calcium channel blocker.1
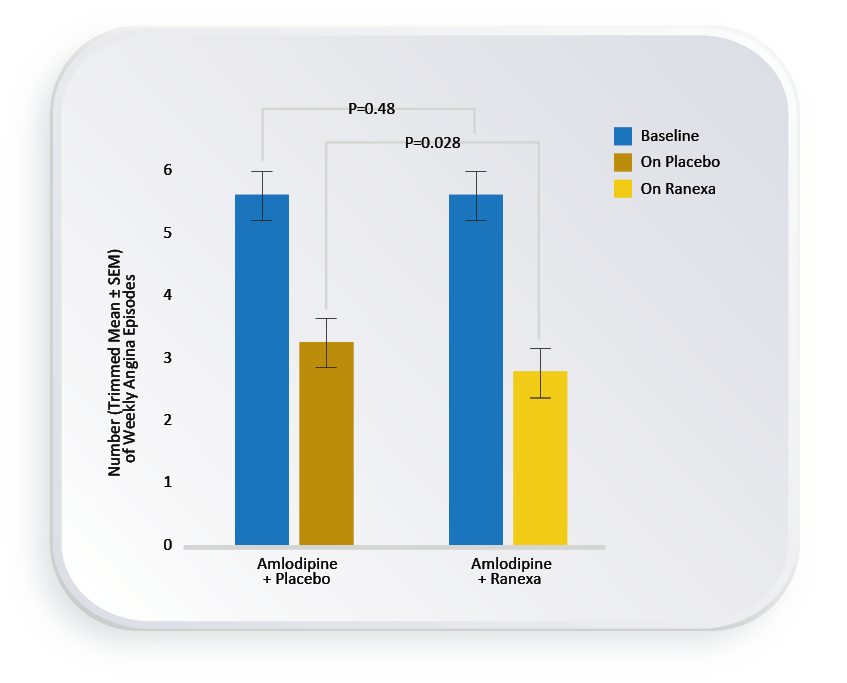

For more details


TERISA study1
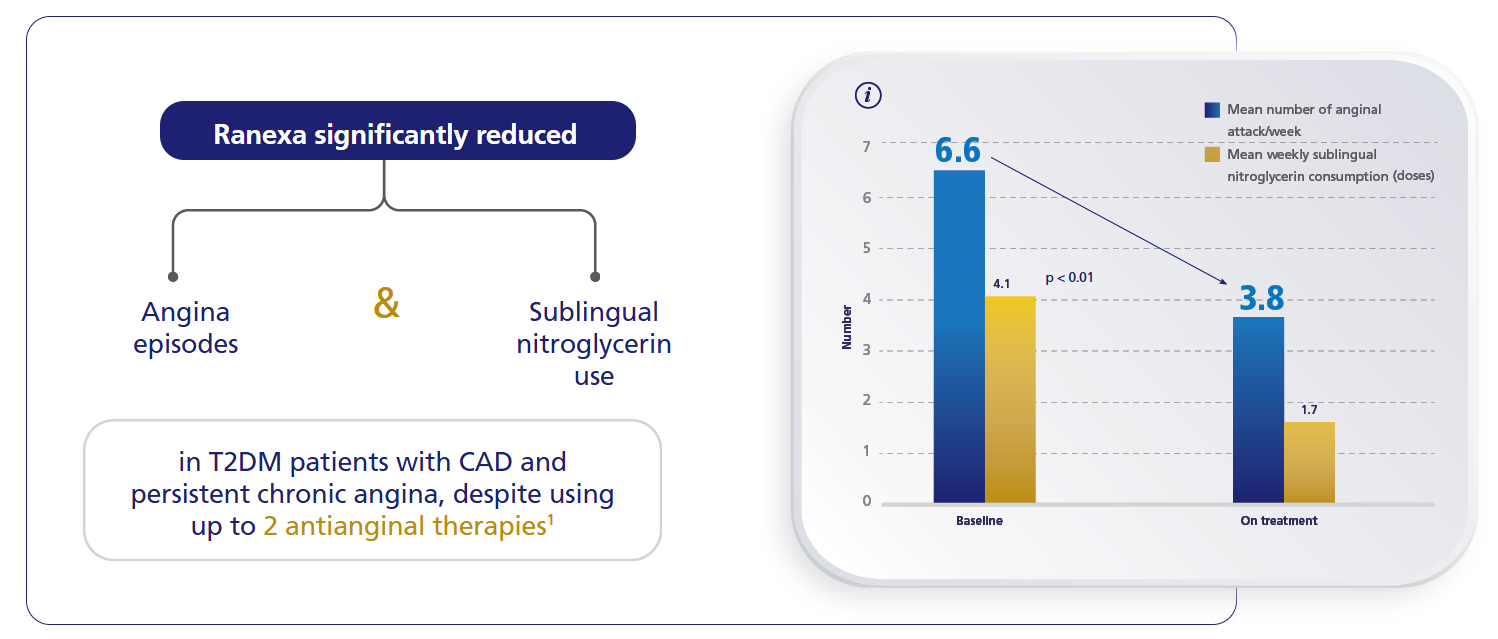

For more details


MERLIN-TIMI 36 Study: Ranexa’s Efficacy in Acute
Coronary Syndrome & Chronic Angina1,2
Ranexa significantly reduces cardiovascular endpoints and angina-related events compared to placebo.

For more details


CARISA Study1
Ranexa significantly increased
treadmill exercise duration by 46.3 Sec at peak drug levels.1 (p=0.006)

For more details


OSCAR Observational Study:
Real-World Evidence for Ranexa1
For more details


For more details


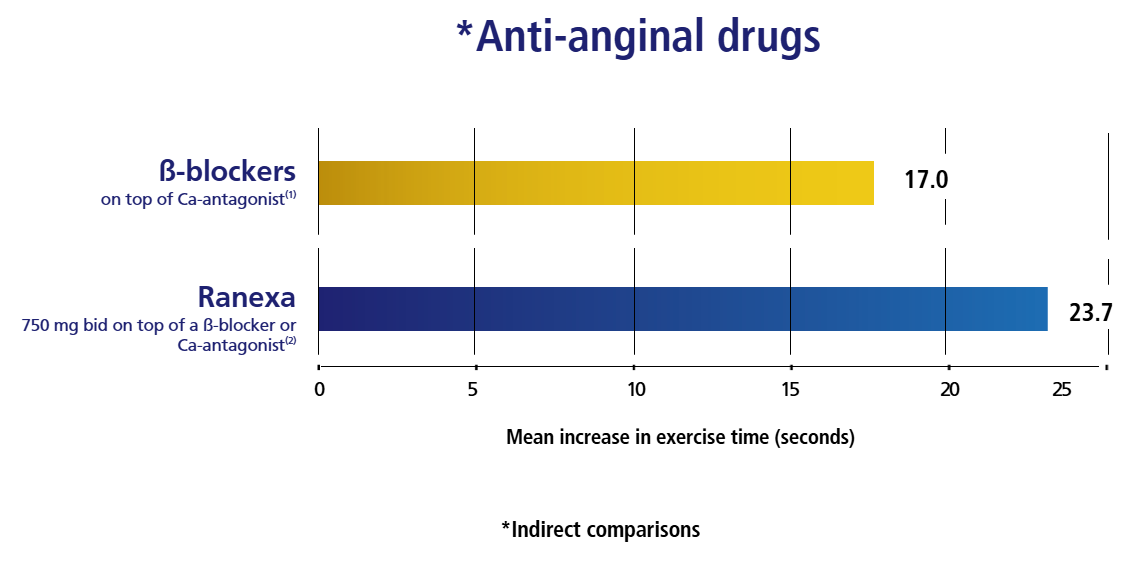
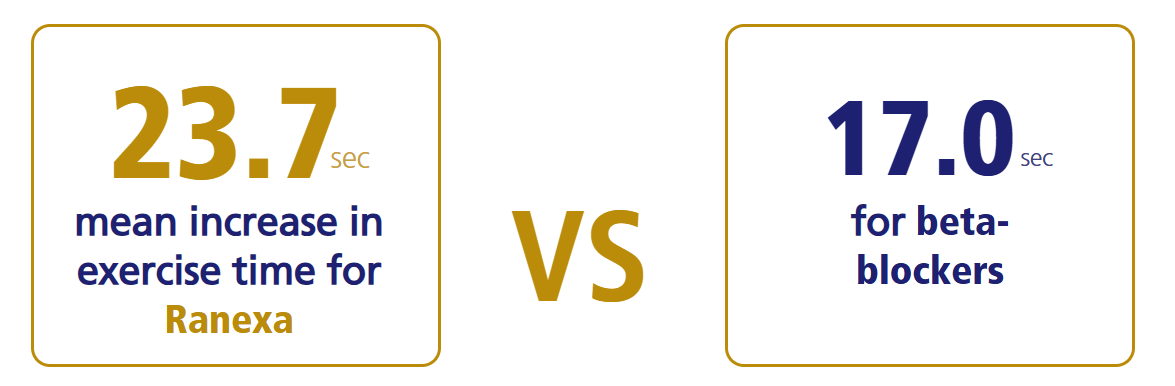
Ranexa VS Beta Blocker1,2
Ranexa improves patient endurance & symptom management with1,2

For more details


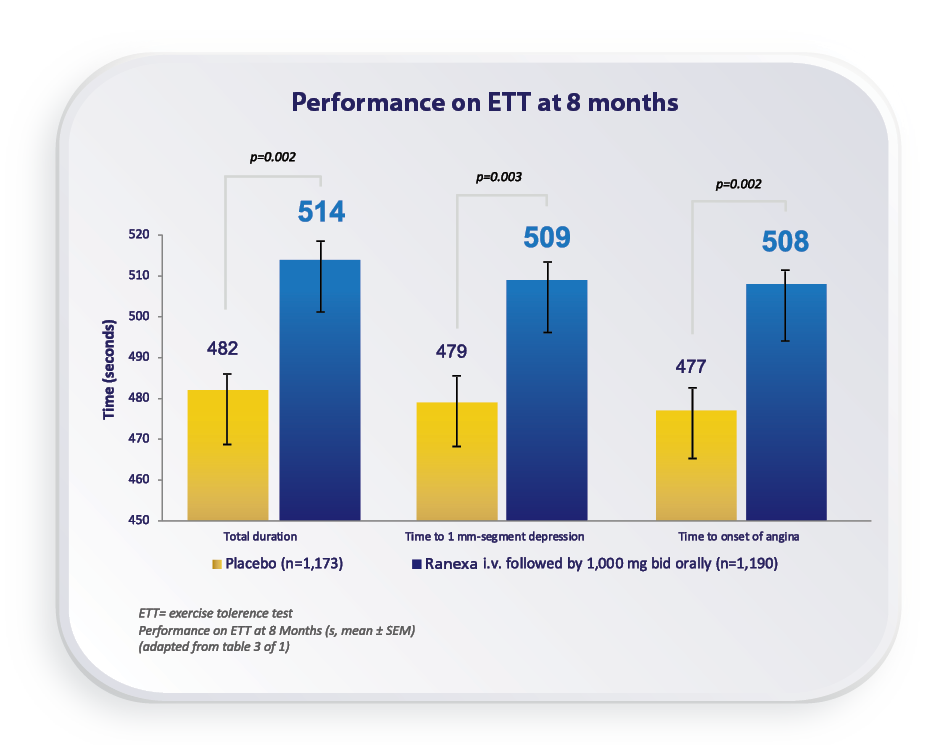
MERLIN-TIMI 36 Study1

Ranexa significantly improved all measures of exercise performance among the patients with ACS and prior chronic angina.1

For more details


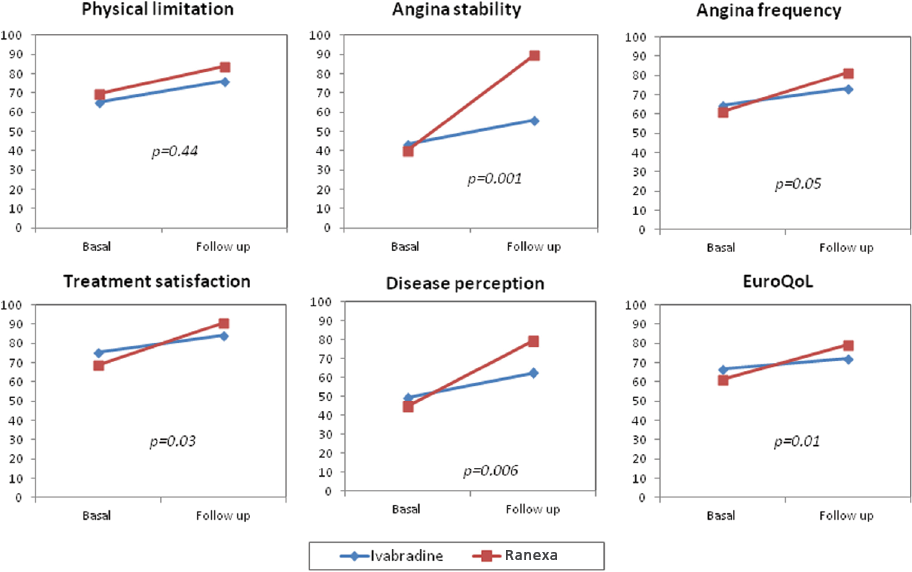
Ranexa Enhances Quality of Life and Symptom Control in Patients with Microvascular Angina.1

For more details


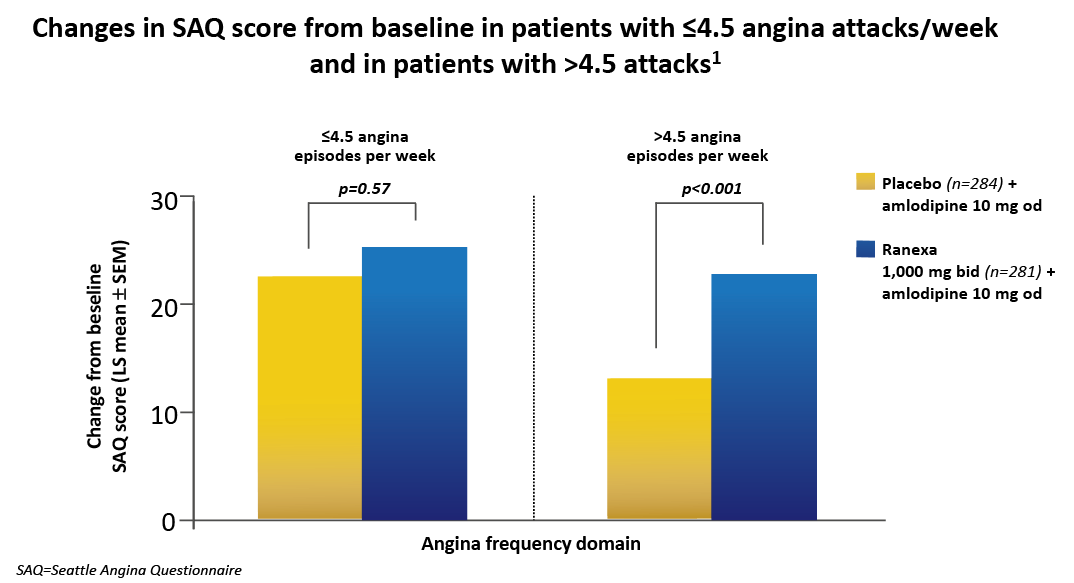
ERICA: Ranexa Significantly
Improves Quality of Life1
Ranexa significantly

for patients with >4.5 angina episodes per week, showing a substantial increase in SAQ scores compared to placebo.1

For more details


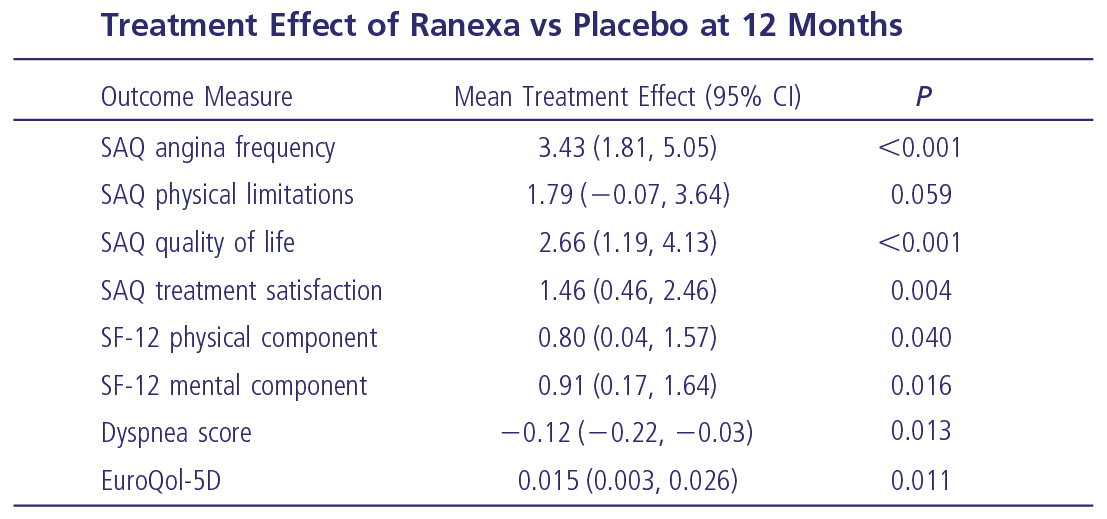
Ranexa Significantly Improves Quality of Life1,2
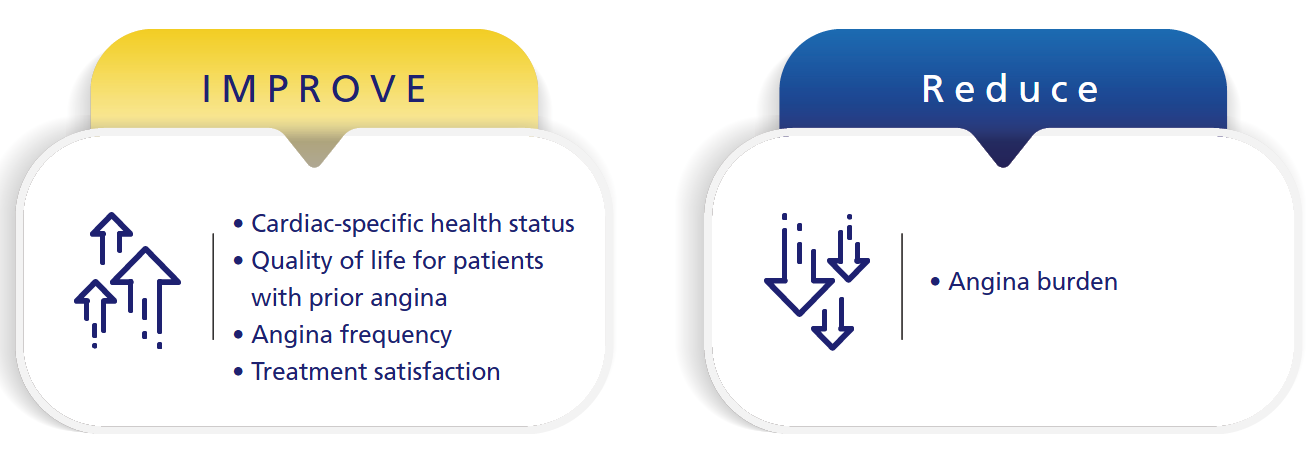

For more details


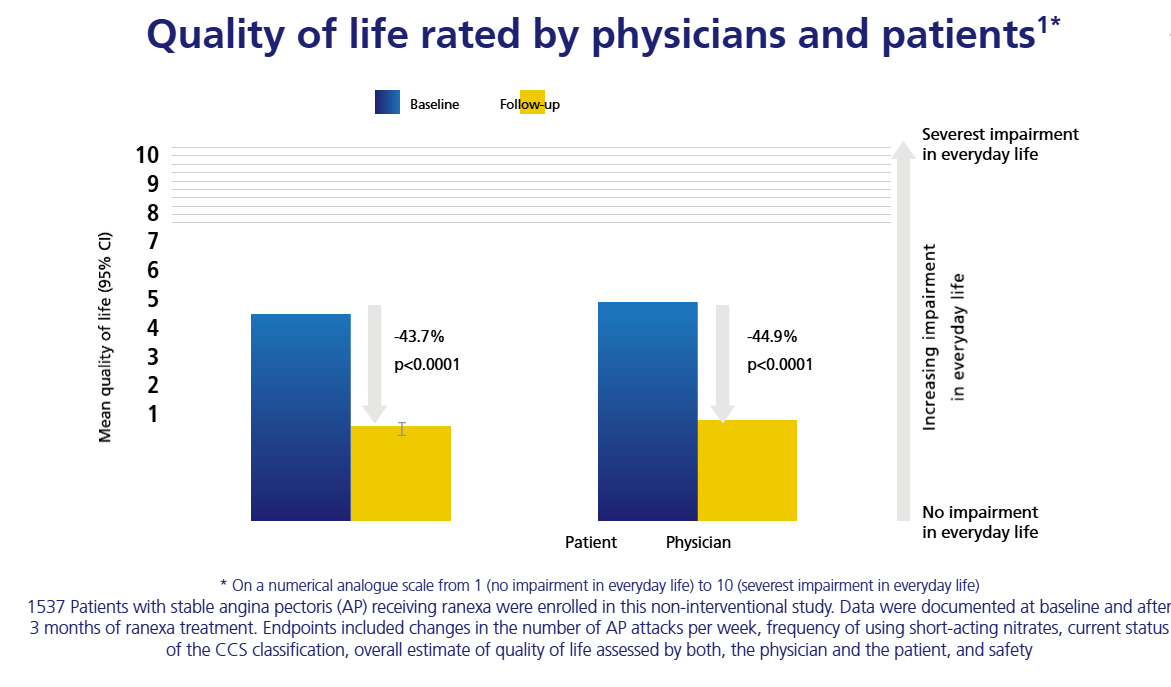
ARETHA Study: Ranexa Transforming Quality of Life in Stable Angina Patients1
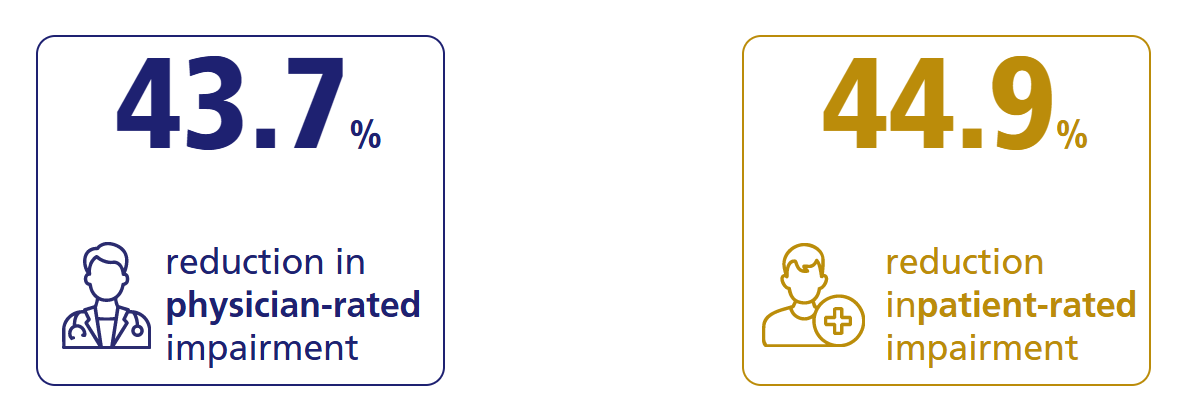

For more details

RANGER Study: The clinical efficacy of Ranexa has been proven in several large randomised, controlled clinical trials that overall enrolled more than 8,000 patients with coronary heart disease1

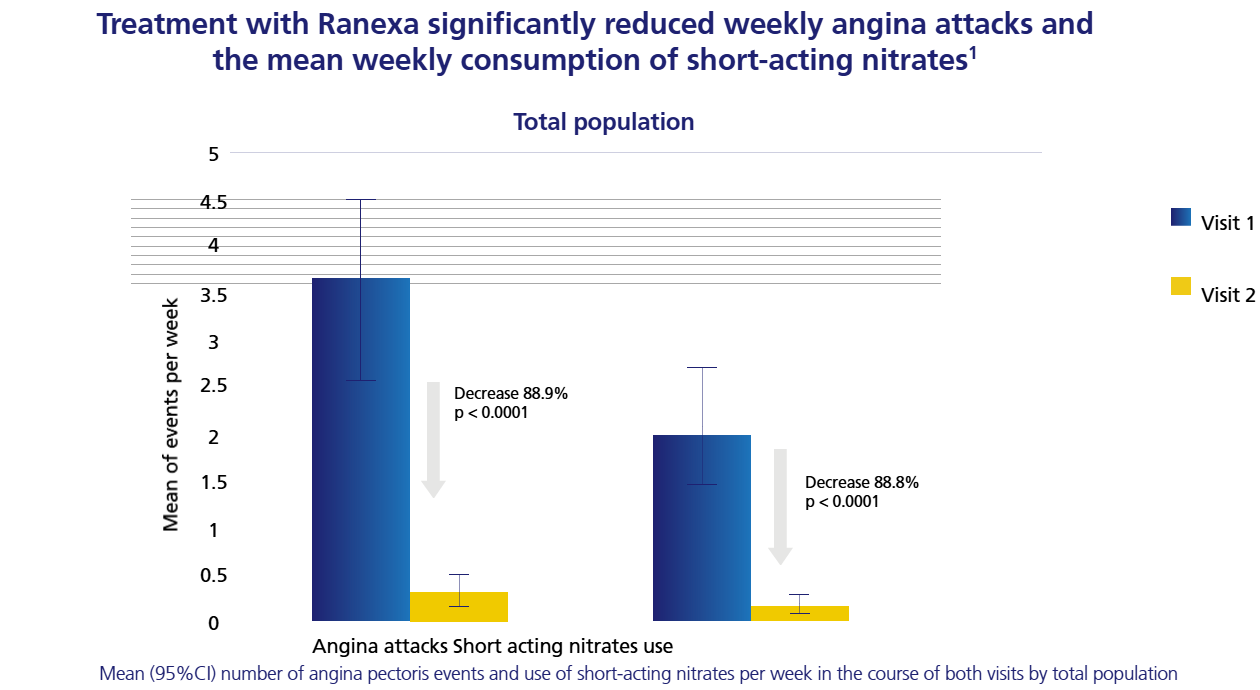
RANGER Study: Real-World Data for Ranexa in Stable Angina1


For more details

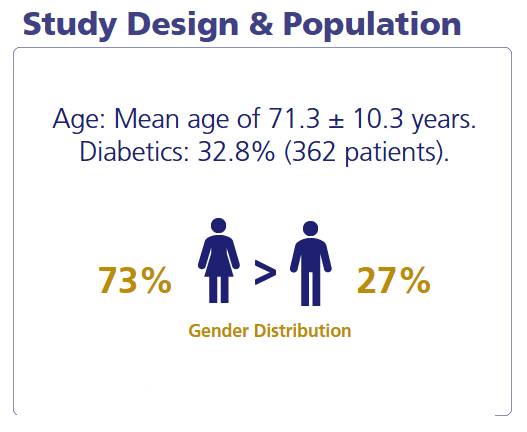

For more details

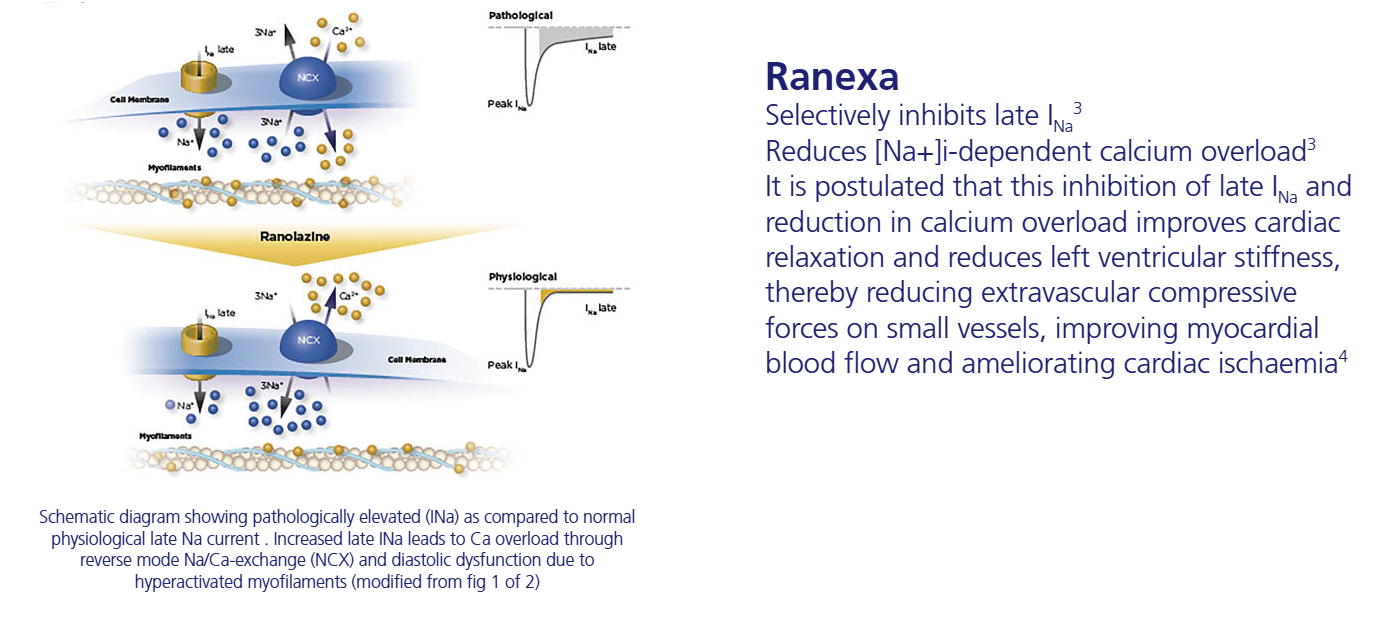
Ranexa Mechanism of Action1
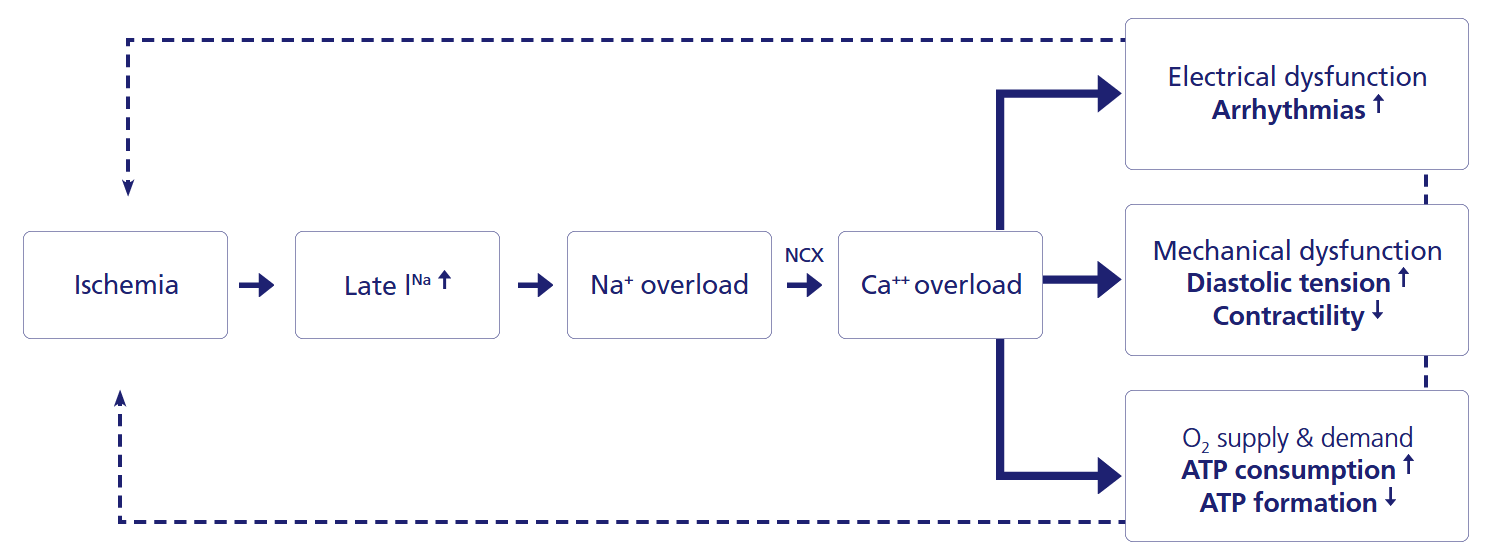

For more details

Ranexa Mechanism of Action1
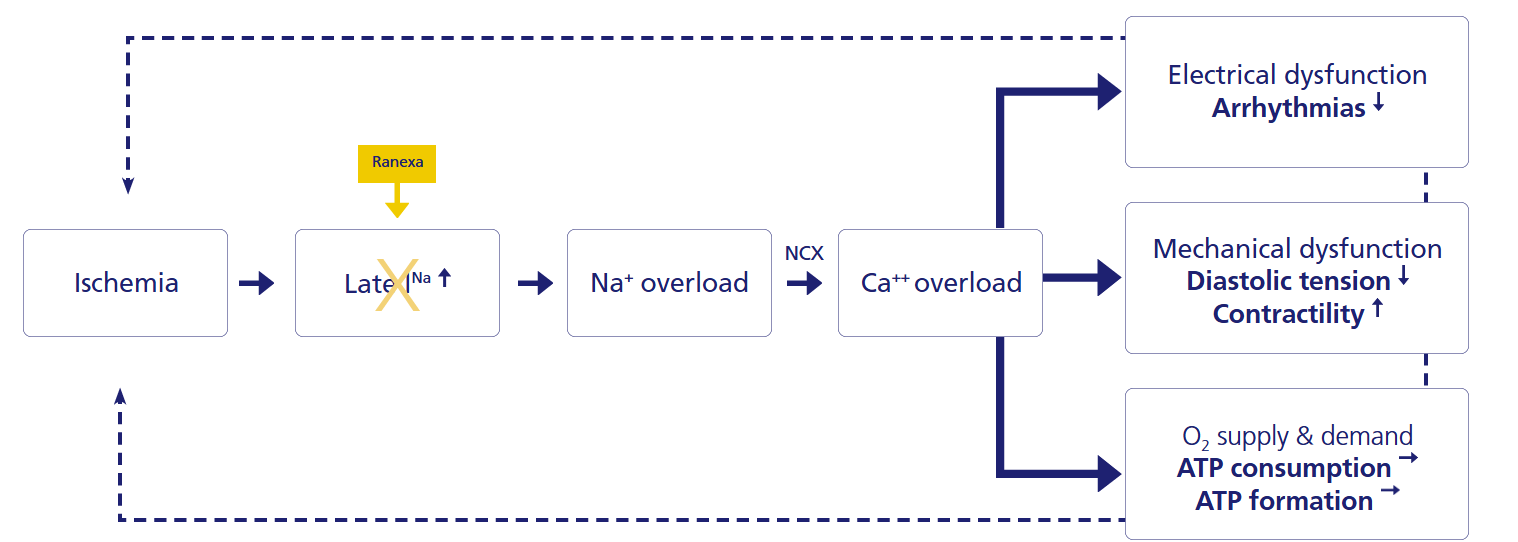

For more details

ESC & ACC/AHA Guidelines Recommendations for Ranexa


For more details



For more details


Tailored Therapeutic Approaches Suggested by Experts1,2
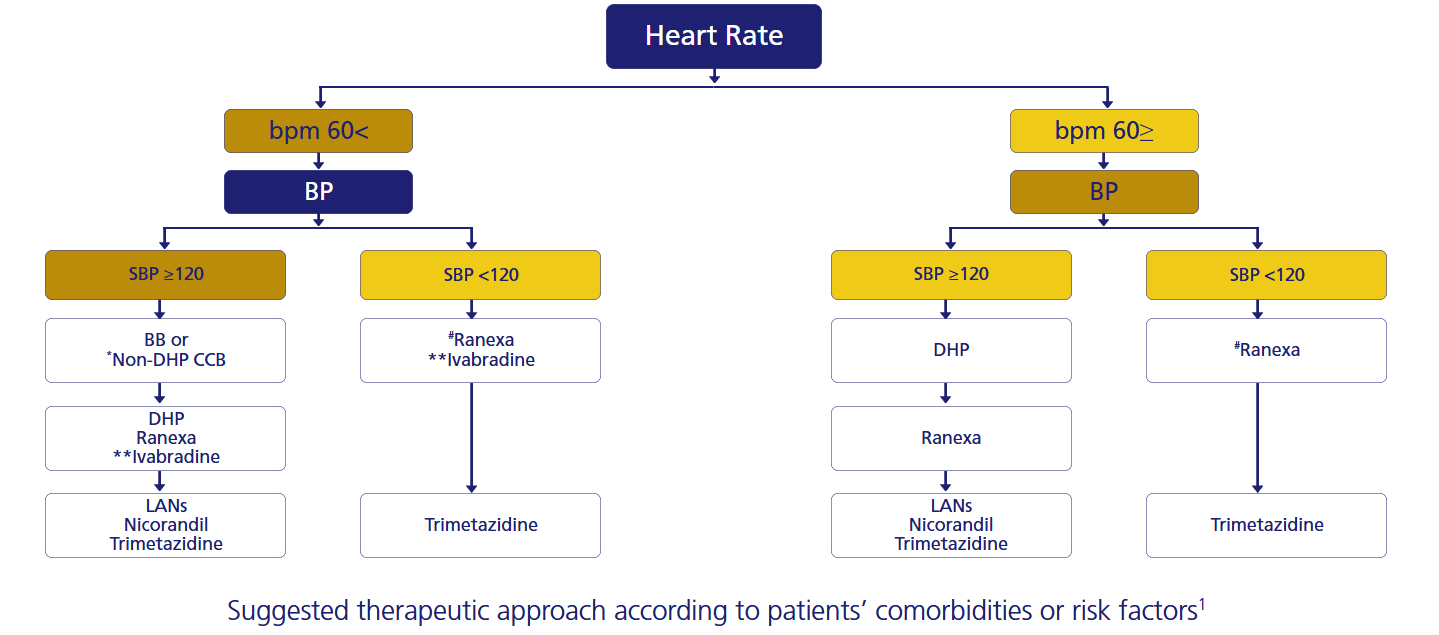

For more details

Choosing Among Antianginals
|
Blood pressure |
Heart rate |
|
|
Long-acting nitrate1 |
|
–— |
|
Ivabradine2 |
–— |
|
|
Nicorandil3 |
|
–— |
|
Diltiazem4 |
|
|
|
Amlodipine5 |
|
–— |
|
Atenolol6 |
|
|
|
Trimetazidine7 |
–— |
–— |
Ranexa effects do NOT depend upon changes in heart rate, blood pressure, or vasodilation.8

Long Acting Nitrates
|
Improvement9 |
ENDOTHELIAL FUNCTION |
Possible worsening10 |
|
No8 |
TOLERANCE |
Yes10 |
|
No8 |
REBOUND EFFECT |
Yes11 |
|
No substantial effect12 |
BLOOD PRESSURE |
Decrease1 |
|
No substantial effect12 |
HEART RATE |
Possible reflex tachycardia1 |

Therapeutic Approach Based on Comorbidities1
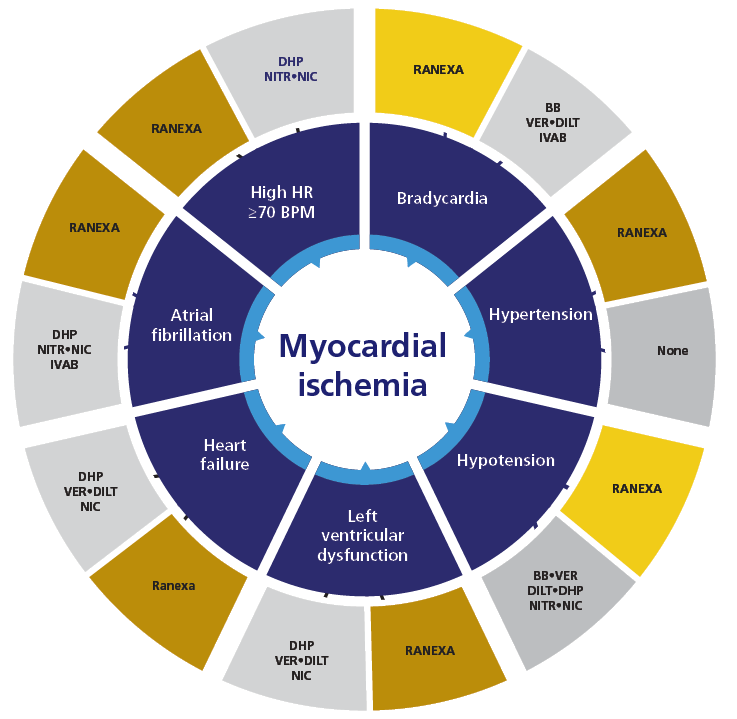


Safety1
Undesirable effects in patients receiving Ranexa® are generally mild to moderate in severity and often develop within the first 2 weeks of treatment. The adverse events reported as common (≥1/100 to <1/10) and considered to be at least possibly related to treatment were: dizziness, headache, constipation, vomiting, nausea and asthenia.1
There is a theoretical risk that concomitant treatment of Ranexa with other drugs known to prolong the QTc interval may give rise to a pharmacodynamic interaction and increase the possible risk of ventricular arrhythmias.
Examples of such drugs include certain antihistamines (e.g. terfenadine, astemizole, mizolastine), certain antiarrhythmics (e.g. quinidine, disopyramide, procainamide), erythromycin, and tricyclic antidepressants (e.g. imipramine, doxepin, amitriptyline).
Main contraindications:
- Severe renal impairment (creatinine clearance < 30 ml/min)
- Moderate or severe hepatic impairment
- Concomitant potent CYP3A4 inhibitors (e.g. itraconazole, ketoconazole, voriconazole, posaconazole, HIV protease inhibitors, clarithromycin, telithromycin, nefazodone)
- Class Ia (e.g. quinidine) or Class III (e.g. dofetilide, sotalol) antiarrhythmics other than amiodarone
Careful dose titration is recommended in:
- Concomitant administration of moderate CYP3A4 inhibitors (e.g. diltiazem, fluconazole, erythromycin)
- Concomitant administration of P-gp inhibitors (e.g. verapamil, ciclosporin)
- Mild hepatic impairment
- Mild to moderate renal impairment (creatinine clearance 30–80 ml/min)
- Elderly
- Patients with low weight (≤ 60 kg)
- Patients with moderate to severe CHF (NYHA Class III–IV)

Posology and Method of Administration1
Ranexa is indicated in adults as add-on therapy for the symptomatic treatment of patients with stable angina pectoris who are inadequately controlled or intolerant to first-line antianginal therapies (such as beta-blockers and/or calcium antagonists)
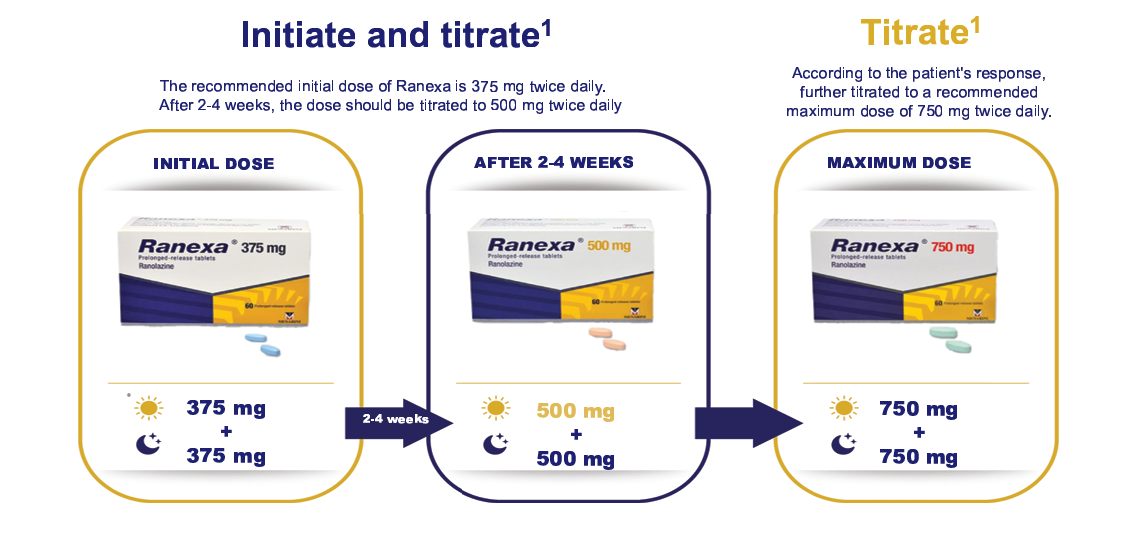

For Ranexa SmPC


For Ranexa SmPC